Beta Elimination Definition:
An elimination reaction in which a proton from the beta (ß) position is removed together with the leaving group, forming a double bond.
Beta Elimination Explained:
Beta elimination, also known as 1,2-elimination, can occur only if the compound consists of a good leaving group. Recall that weak bases are almost always good leaving groups. Some examples of weak bases include:
(1) Halide Ions: I-, Br-, Cl- (Reaction called: Dehydrohalogenation)
(2) Water (Reaction called: Dehydration)
(3) p-toluenesulfonate
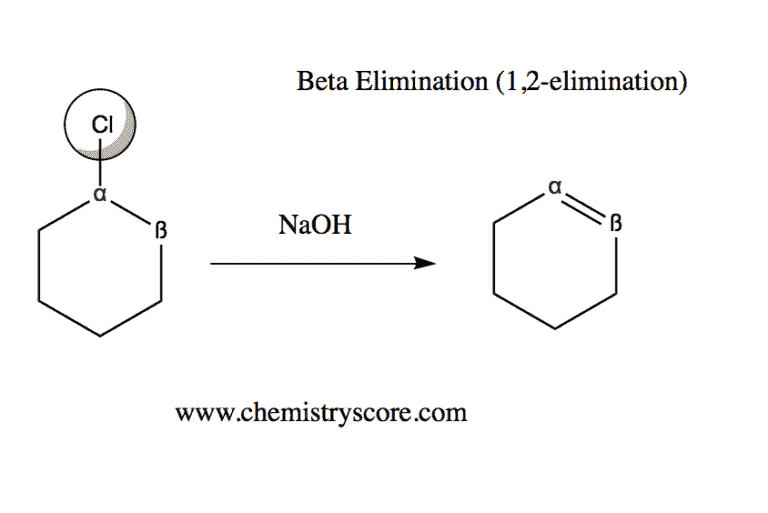
The key point in this type of elimination is to recognize that the end product results in a double bond [C=C], which is called an alkene.