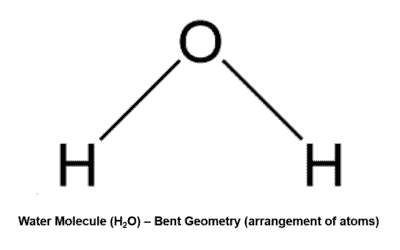
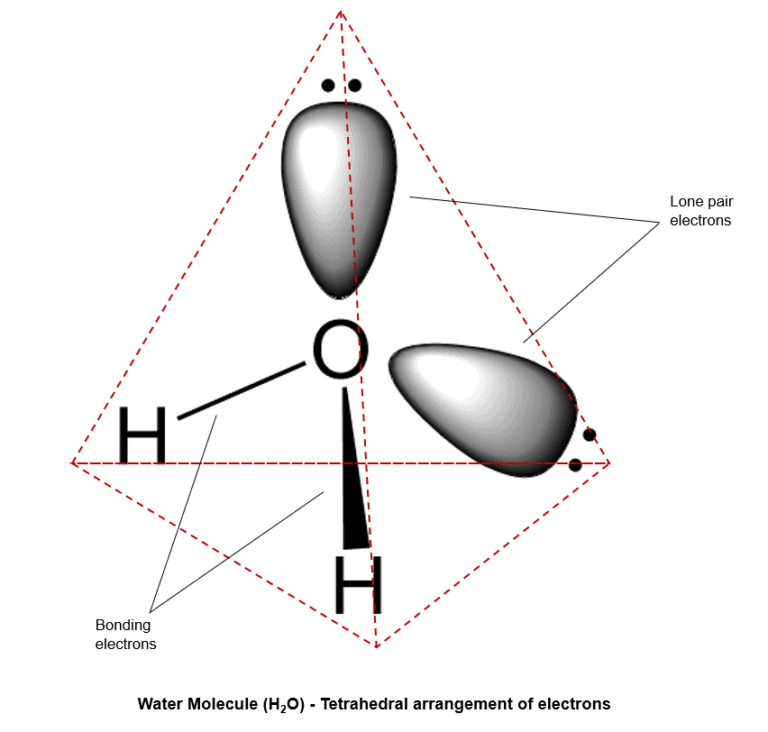
Bent Definition:
The Bent geometry, is a type of atomic arrangement in molecules resulting from an sp3-hybridized atom that has two lone pairs.
Bent Explanation:
When we hear the word geometry, it should be clear to us that geometry refers to the arrangement of atoms in a molecule and does not refer to the arrangement of electron pairs.
In the context of an sp3-hybridized atom, the arrangement of electrons is almost always tetrahedral because of the presence of 4 sp3-hybridized orbitals (steric number 4). However, the arrangement of atoms, depending upon the number of lone pair electrons, can be a Tetrahedral (eg: CH4, no lone pairs), Trigonal Pyramidal (eg: NH2, one lone pair) or a Bent geometry (eg: H2O, two lone pairs)
Example – Water Molecule