Beer’s Law Definition:
In UV-Vis spectroscopy, the amount of UV light absorbed at λmax (lambda max) is given by the equation describing the relationship between molar absorptivity (e), absorbance (A), concentration (C) in (mol/L), and path length (l ):
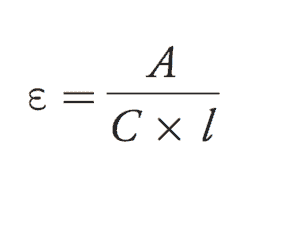
Beer’s Law Explained:
λmax is dependent on the amount of conjugation in a particular organic molecule. The more highly conjugated a compound, the less energy it requires to elicit excitation. Recall the formula:
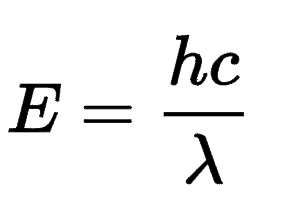
From the equation, you can deduce that energy (E) is inversely proportional to wavelength (λ). Thus, longer wavelengths correspond to lower energy and vice versa. We know that the more conjugated a compound, i.e. more double and triple bonds, the longer the wavelength, and thus the lower the energy.