Base Peak Definition:
The definition of the base peak, in mass spectroscopy, is a relative value of 100% assigned to the tallest peak in a given spectrum
Base Peak Explained:
In the ionization process for mass spectroscopy, the positive ions are separated by their mass-to-charge ratio (m/z). Because the charge for most ions is +1, m/z, effectively becomes a measure of the mass of the individual positive ion. The plot generated is called the mass spectrum, the tallest peak in which is called the base peak – the height of every other peak is described relative to the height of the base peak. The base peak, hence, represents the most abundant positive ion formed during the spectroscopy.
Consider the break up for the following molecular ion M
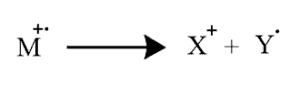
Here, the uncharged free radical Y does not produce lines on the mass spectrum, and only the charged particle X is detected by the spectrometer and produces the spectrum. For complex molecules, various fragmentations of the molecular ion take place, but the one with the highest abundance produces the tallest spectrum line.
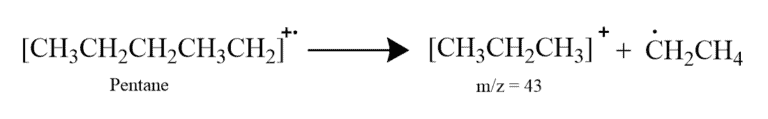
For Example – In the mass spectrum for pentane, the positive ion with m/z = 43 produces the tallest spectrum. This corresponds one of the possible fragmentations where pentane breaks up as