Aufbau Principle Definition:
The Aufbau principle is a rule governing how the electrons are assigned to atomic orbitals. It states that the lowest energy orbital is filled first.
Aufbau Principle Explanation:
On a broader level, there are three principles to be followed when assigning electrons to an atom. The Aufbau principle, The Hund’s rule, and The Pauli’s Exclusion principle. Out of these, the Aufbau principle is the starting point. The principle essentially explains that a when occupying orbitals and electron has the tendency to do so by occupying the orbital with the lowest energy level first. i.e. an electron will not occupy the 2s orbital before the 1s orbital is completely filled. To understand how electrons are assigned in individual orbitals we must follow the other rules mentioned.
Example –
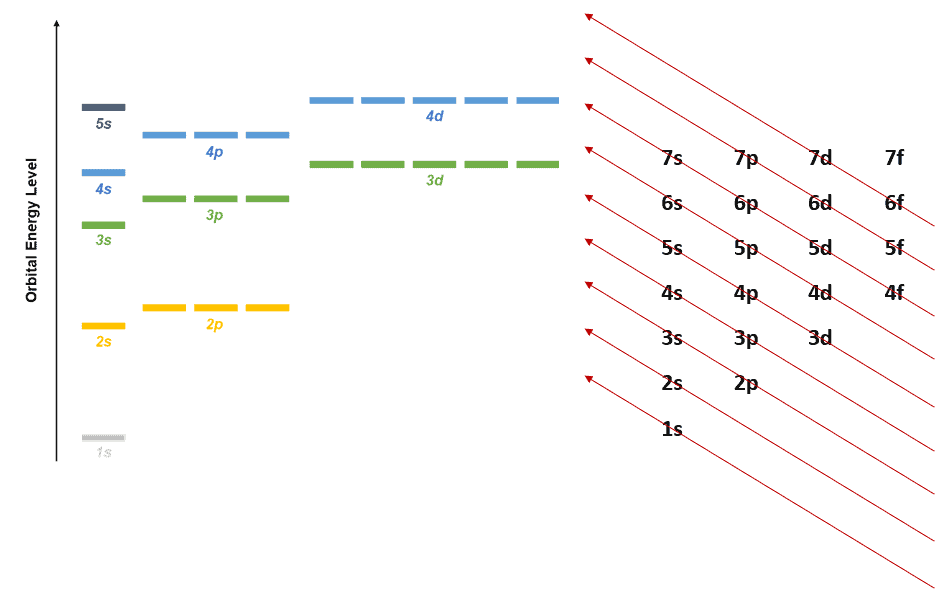
The diagram on the left represents the atomic orbitals by energy level. The diagram on the right is the representation of the Aufbau principle that shows how electrons fill up the orbitals. You should traverse the arrows starting from the lowest and move up along the direction in which the arrows are pointing. eg – from 1s -> 2s -> 2p -> 3s -> 3p -> 4s -> 3d -> 4p and so on