Antiaromatic Definition:
Instability that arises when a planar ring of continuously overlapping p orbitals contains [4n] Π electrons.
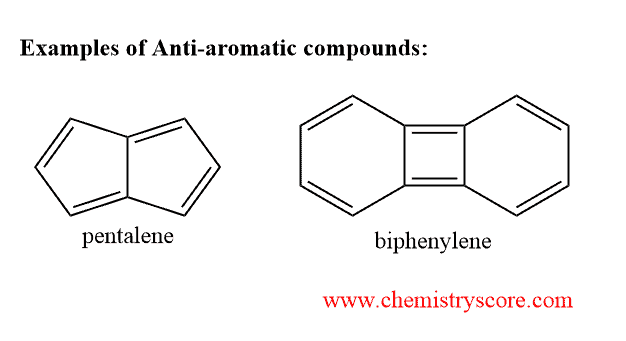
Antiaromatic Explained:
Hückel’s rule states that a compound is aromatic if it follows the [4n + 2] rule. This means that any compound with 2,6,10,14,18… Π electrons, then it is aromatic. However, for anti-aromatic compounds, the rule is slightly different. Compounds that have [4n] Π electrons are said to be anti-aromatic.
Take a look at pentalene above. It has 4 Π bonds within the structure. If you apply the 4n rule, you get:
4 X 4 (since n = 4 pi bonds) = 16; therefore pentalene is antiaromatic