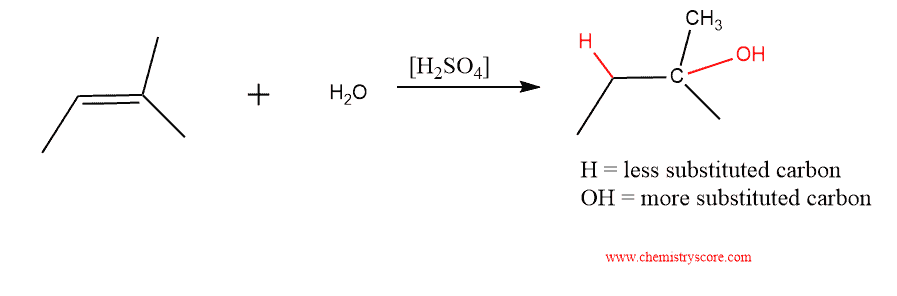
Acid-Catalyzed Hydration Definition:
A reaction that achieves the addition of water across a double bond in the presence of an acid catalyst.
Acid-Catalyzed Hydration Explained:
The addition of water across a double bond with an acid catalyst proceeds via the markovnikov addition pathway. The result is an addition of hydrogen (H) and alcohol (OH) across the double bond. The alcohol (OH) group is added to the more substituted carbon. The diagram below depicts the acid-catalyzed hydration reaction scheme: