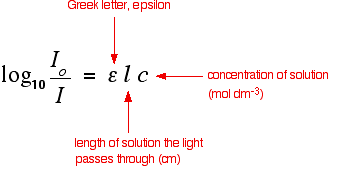
Absorbance Definition (Chemistry):
n UV-Vis spectroscopy, the value log (I0 /I) where I0 is the intensity of the reference beam and I is the intensity of the sample beam.
Absorbance In Chemistry Explained:
In chemistry, spectroscopy allows us to understand molecular structures by analyzing the interaction between matter and electromagnetic radiation. In UV-vis spectroscopy, absorbance is determined from two data sets. The first data set is the wavelength (ranging from 200 to 800 nm) of a beam passing through a cuvette (small glass container) containing the organic material dissolved in appropriate solvent. The second data set is the wavelength (again, ranging from 200 to 800 nm) containing only the solvent in a cuvette, acting as a “reference”. The I0 of the absorbance formula is the intensity (in nm) of the “reference” and the I is the intensity of the sample.