Dihydroxylation [OsO4]
Dihydroxylation Definition:
When an alkene is treated with osmium tetroxide (OsO4), two hydroxyl groups are added to the alkene by cleaving the double bond. This process is called syn dihydroxylation.
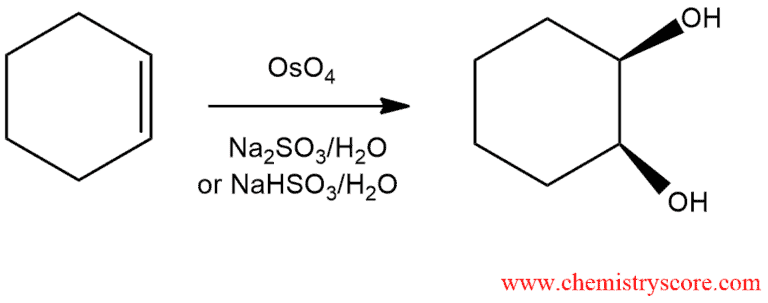
Dihydroxylation Explained:
In this process, which consists of two phases, the corresponding vicinal diols in a stereospecifically syn manner are obtained. Syn means that the product can form only in a way that introduces the two oxygen atoms on the same face of the double bond.
In the first phase, an alkene is treated with osmium tetroxide (OsO4), and a cyclic osmate ester is produced. This initial reaction of the π bond with osmium tetroxide constitutes a concerted addition in which three electron pairs move simultaneously. This intermediate is usually not isolated but converted upon reductive work-up into the free diol.
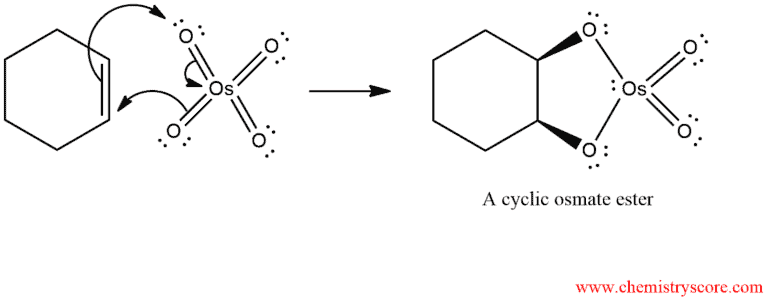
In the second phase, the cyclic ester of osmium is treated with aqueous sodium sulfite (Na2SO3) or sodium bisulfite (NaHCO3) to give a diol:
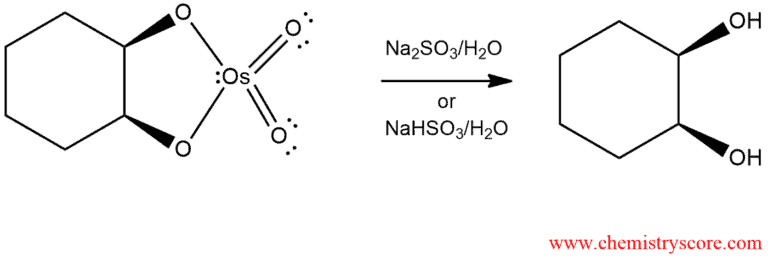
This method can be used to convert alkanes into diols with fairly high yields, but there are several disadvantages. Osmium tetroxide is expensive and highly toxic. To deal with these issues, several methods have been developed that use a co-oxidant that serves to regenerate OsO4 as it is consumed during the reaction. In this way, osmium tetroxide functions as a catalyst so that even small quantities can produce large quantities of the diol.