Resonance Stabilization Definition:
Resonance stabilization is the stabilization offered due to the delocalization of electrons in a molecule.
Resonance Stabilization Explained:
Resonance happens in the initiation of delocalization in which the overall energy of an atom is lowered since its electrons occupy a greater volume. Therefore, the molecules that experience resonance are more stable than those that do not and are termed resonance stabilized.
Example –
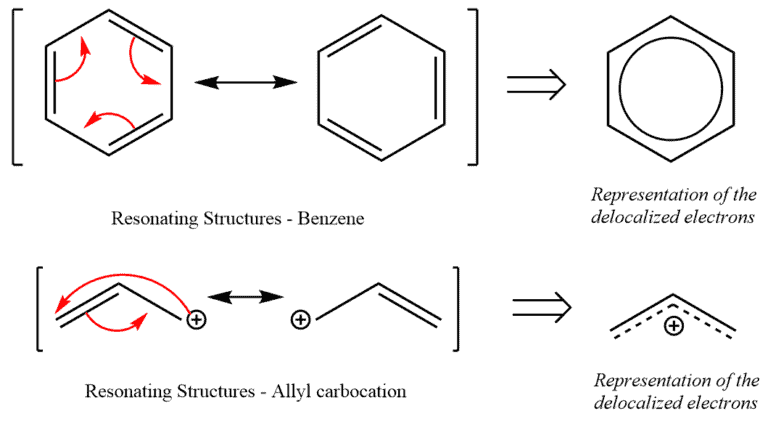
Notice the spreading of charge. This stabilizes the molecule as the overall energy of the molecule reduces.