Delocalized Definition:
Delocalized, is the term given to the lone pair of electrons that participate in resonance.
Delocalized Explained:
A delocalized electron or electron pair is a freely moving electron pair that is responsible for different resonating structures. Since they take part in resonance it is important to note that the delocalized pair of electrons always occupy a standalone p orbital and not the hybridized orbital. Also, see Delocalization.
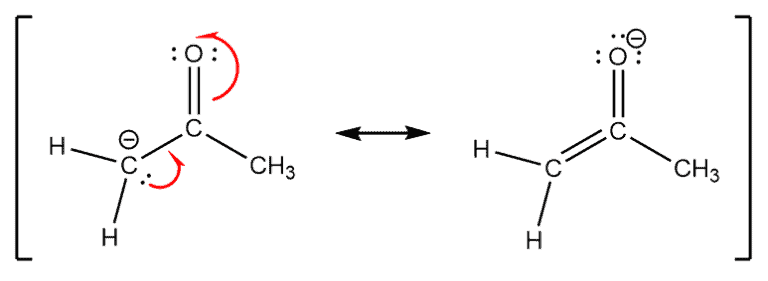
Example –
Consider the resonating structures for the Acetone anion. Notice that the carbon atom is sp2 hybridized and has the delocalized pair of electrons in a p orbital. This enables the formation of the pi bond in the structure on the right-hand side.