Alkyl iodide formation [HI]
Alkyl iodide formation (HI) Definition:
Tertiary alcohol can be converted to tertiary alkyl iodide with hydroiodic acid (HI).
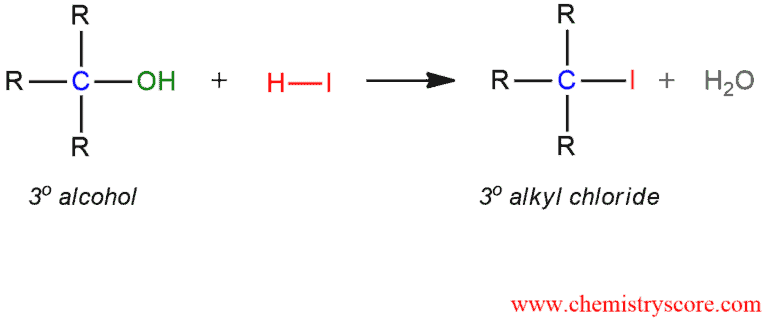
Alkyl iodide formation (HI) Explained:
This reaction proceeds via an SN1 mechanism which consists of three steps:
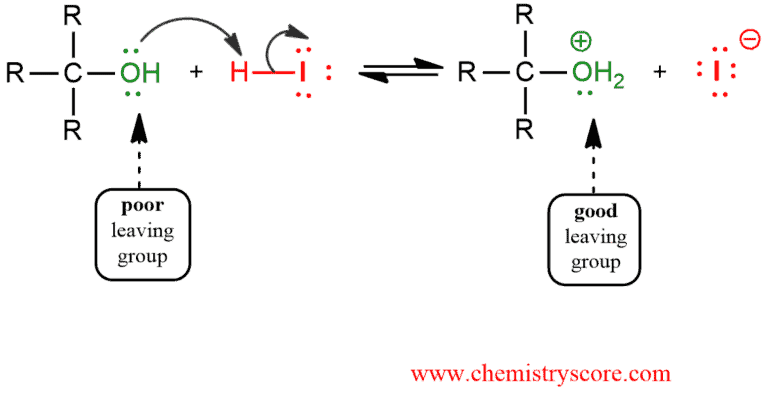
1. An acid/base reaction. Protonated oxygen from the hydroxyl group of alcohol makes a better leaving group.
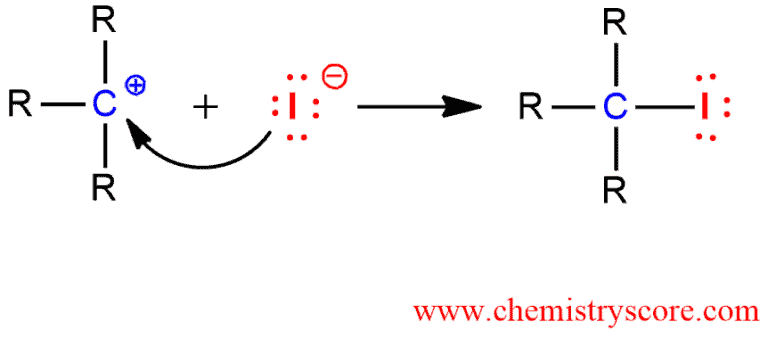
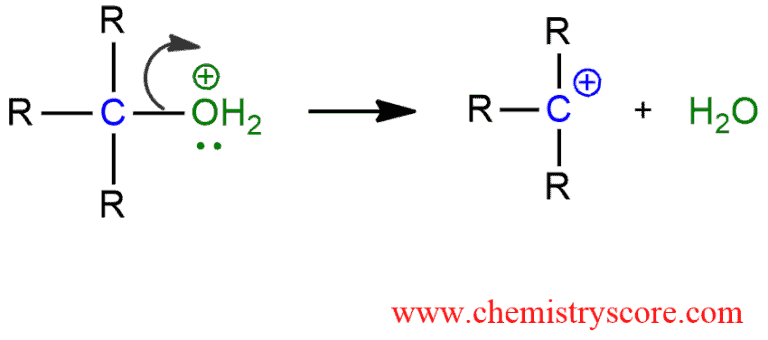
2. The rate-determining step. Loss of the leaving group and carbocation formation.
3. Nucleophilic attack of iodide ion on the electron-deficient carbon make final product (alkyl iodide)